Life: Magnified
Snapshots of Life: Cell Skeleton on the Move
Posted on by Dr. Francis Collins
Cells are constantly on the move. They shift, grow, and migrate to new locations—for example, to heal a wound or to intercept an infectious agent as part of an immune response. But how do cells actually move?
In this image, Torsten Wittmann, an NIH-funded cell biologist at the University of California, San Francisco, reveals the usually-invisible cytoskeleton of a normal human skin cell that lends the cell its mobility. The cytoskeleton is made from protein structures called microtubules—the wispy threads surrounding the purple DNA-containing nucleus—and filaments of a protein called actin, seen here as the fine blue meshwork in the cell periphery. Both actin and microtubules are critical for growth and movement.
Snapshots of Life: Visualizing Blood Vessels
Posted on by Dr. Francis Collins
This might look a bit like a fish net, but what’s actually caught in this image is the structure of the endothelium—the thin layer of cells lining your blood vessels that controls the flow of molecules in and out of the bloodstream. The red lines are the actin filaments that give each endothelial cell its shape, while the purple are proteins called cadherins.
Most of the time, the actin “ropes” and cadherin “glue” act together to form a tight seal between endothelial cells, ensuring that nothing leaks out of blood vessels into surrounding tissue. However, when endothelial cells sense an infection or an injury, the cadherins open gaps that allow various disease-fighting or healing factors or cells present in the blood to breach the barrier and enter infected or injured tissue. After the infection subsides or wound heals, the gaps close and the blood vessel is once again impenetrable.
Snapshots of Life: Fat (Tissue) Is Beautiful
Posted on by Dr. Francis Collins
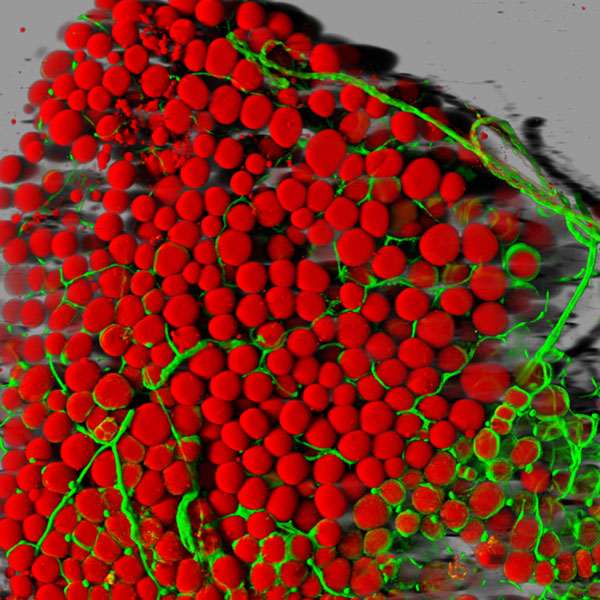
Caption: Fat cells (red) surrounded by blood vessels (green) that supply them with nutrients.
Credit: Daniela Malide, National Heart, Lung, and Blood Institute; NIH
With all of today’s sophisticated microscopes, you’d think it would be simple to take high-magnification photos of fat—but it’s not. Fat tissue often leaks slippery contents, namely lipids, when it’s thinly sliced for viewing under a microscope. And even when a sample is prepared without leakage, there’s another hurdle: the viscous droplets of lipid contained in the fat cells block light from passing through.
So, it’s good news that one of NIH’s intramural scientists here in Bethesda, MD, has come up with a way to produce high-resolution, 3-D images of fat cells like the one you see above. Not only are these images aesthetically appealing, but they’ll be valuable to efforts to expand our understanding of this essential and much-maligned tissue.
Snapshots of Life: Portrait of Skin Cancer
Posted on by Dr. Francis Collins
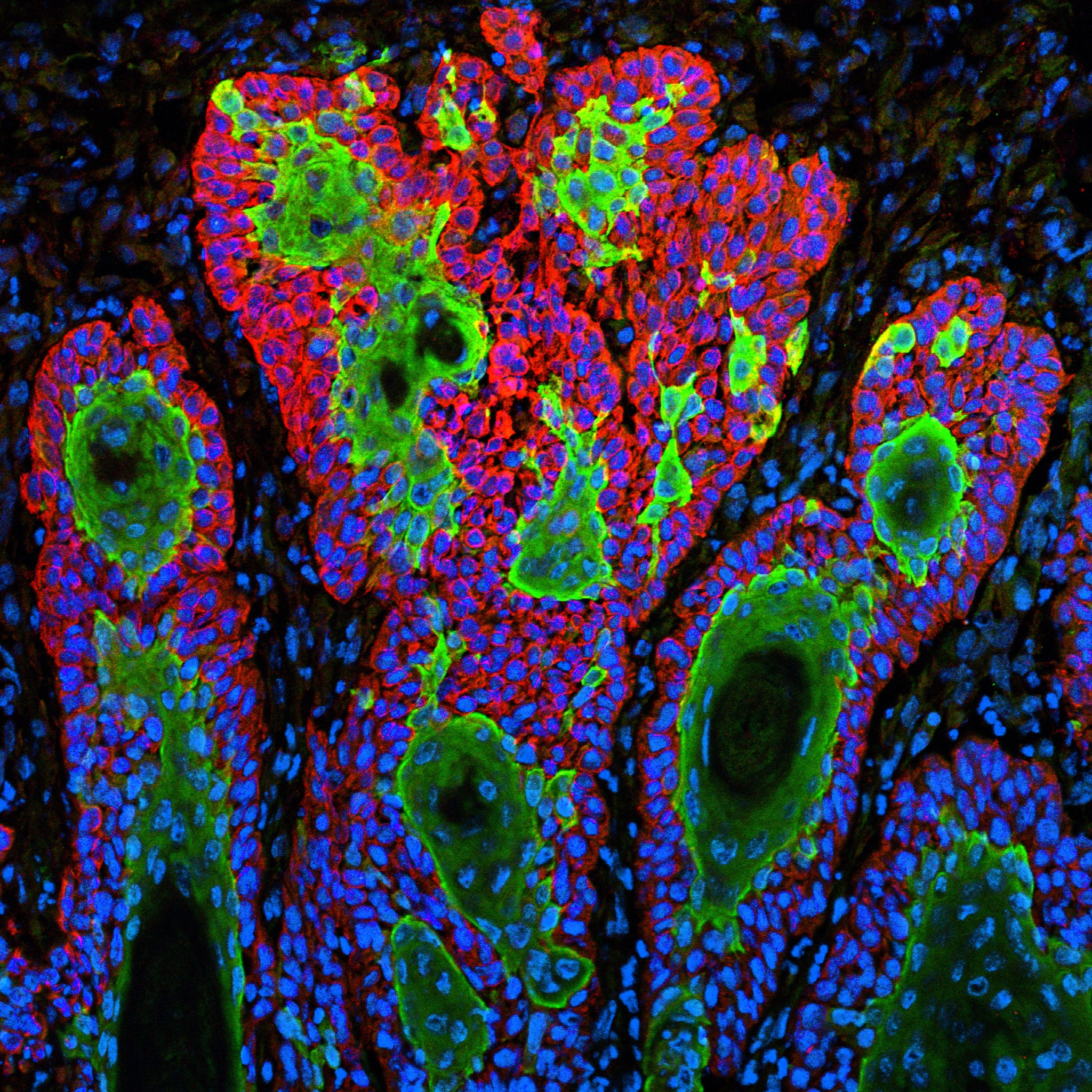
Caption: This image shows the uncontrolled growth of cells in squamous cell carcinoma.
Credit: Markus Schober and Elaine Fuchs, The Rockefeller University, New York
For Markus Schober, science is more inspiring when the images are beautiful, even when the subject is not. So, when this biologist was at The Rockefeller University in New York and peered through his microscope at squamous cell carcinoma (SCC), both the diabolical complexity—and the beauty—of this common form of skin cancer caught his eye.
Schober wasn’t the only one who found the image compelling. A panel of judges from the National Institute of General Medical Sciences and the American Society for Cell Biology chose to feature it in their Life: Magnified exhibit, which recently opened at the Washington Dulles International Airport.




