non-small cell lung cancer
New Tool Predicts Response to Immunotherapy in Lung Cancer Patients
Posted on by Douglas M. Sheeley, Sc.D., NIH Common Fund

With just a blood sample from a patient, a promising technology has the potential to accurately diagnose non-small cell lung cancer (NSCLC), the most-common form of the disease, more than 90 percent of the time. The same technology can even predict from the same blood sample whether a patient will respond well to a targeted immunotherapy treatment.
This work is a good example of research supported by the NIH Common Fund. Many Common Fund programs support development of new tools that catalyze research across the full spectrum of biomedical science without focusing on a single disease or organ system.
The emerging NSCLC prediction technology was developed as part of our Extracellular RNA Communication Program. The program develops technologies to understand RNA circulating in the body, known as extracellular RNA (exRNA). These molecules can be easily accessed in bodily fluids such as blood, urine, and saliva, and they have enormous potential as biomarkers to better understand cancer and other diseases.
When the body’s immune system detects a developing tumor, it activates various immune cells that work together to kill the suspicious cells. But many tumors have found a way to evade the immune system by producing a protein called PD-L1.
Displayed on the surface of a cancer cell, PD-L1 can bind to a protein found on immune cells with the similar designation of PD-1. The binding of the two proteins keeps immune cells from killing tumor cells. One type of immunotherapy interferes with this binding process and can restore the natural ability of the immune system to kill the tumor cells.
However, tumors differ from person to person, and this form of cancer immunotherapy doesn’t work for everyone. People with higher levels of PD-L1 in their tumors generally have better response rates to immunotherapy, and that’s why oncologists test for the protein before attempting the treatment.
Because cancer cells within a tumor can vary greatly, a single biopsy taken at a single site in the tumor may miss cells with PD-L1. In fact, current prediction technologies using tissue biopsies correctly predict just 20 – 40 percent of NSCLC patients who will respond well to immunotherapy. This means some people receive immunotherapy who shouldn’t, while others don’t get it who might benefit.
To improve these predictions, a research team led by Eduardo Reátegui, The Ohio State University, Columbus, engineered a new technology to measure exRNA and proteins found within and on the surface of extracellular vesicles (EVs) [1]. EVs are tiny molecular containers released by cells. They carry RNA and proteins (including PD-L1) throughout the body and are known to play a role in communication between cells.
As the illustration above shows, EVs can be shed from tumors and then circulate in the bloodstream. That means their characteristics and internal cargo, including exRNA, can provide insight into the features of a tumor. But collecting EVs, breaking them open, and pooling their contents for assessment means that molecules occurring in small quantities (like PD-L1) can get lost in the mix. It also exposes delicate exRNA molecules to potential breakdown outside the protective EV.
The new technology solves these problems. It sorts and isolates individual EVs and measures both PD-1 and PD-L1 proteins, as well as exRNA that contains their genetic codes. This provides a more comprehensive picture of PD-L1 production within the tumor compared to a single biopsy sample. But also, measuring surface proteins and the contents of individual EVs makes this technique exquisitely sensitive.
By measuring proteins and the exRNA cargo from individual EVs, Reátegui and team found that the technology correctly predicted whether a patient had NSCLC 93.2 percent of the time. It also predicted immunotherapy response with an accuracy of 72.2 percent, far exceeding the current gold standard method.
The researchers are working on scaling up the technology, which would increase precision and allow for more simultaneous measurements. They are also working with the James Comprehensive Cancer Center at The Ohio State University to expand their testing. That includes validating the technology using banked clinical samples of blood and other bodily fluids from large groups of cancer patients. With continued development, this new technology could improve NSCLC treatment while, critically, lowering its cost.
The real power of the technology, though, lies in its flexibility. Its components can be swapped out to recognize any number of marker molecules for other diseases and conditions. That includes other cancers, neurodegenerative diseases, traumatic brain injury, viral diseases, and cardiovascular diseases. This broad applicability is an example of how Common Fund investments catalyze advances across the research spectrum that will help many people now and in the future.
Reference:
[1] An immunogold single extracellular vesicular RNA and protein (AuSERP) biochip to predict responses to immunotherapy in non-small cell lung cancer patients. Nguyen LTH, Zhang J, Rima XY, Wang X, Kwak KJ, Okimoto T, Amann J, Yoon MJ, Shukuya T, Chiang CL, Walters N, Ma Y, Belcher D, Li H, Palmer AF, Carbone DP, Lee LJ, Reátegui E. J Extracell Vesicles. 11(9):e12258. doi: 10.1002/jev2.12258.
Links:
Video: Unlocking the Mysteries of Extracellular RNA Communication (Common Fund)
Extracellular RNA Communication Program (ERCC) (Common Fund)
Upcoming Meeting: ERCC19 Research Meeting (May 1-2, 2023)
Eduardo Reátegui Group for Bioengineering Research (The Ohio State University College of Engineering, Columbus)
Note: Dr. Lawrence Tabak, who performs the duties of the NIH Director, has asked the heads of NIH’s Institutes, Centers, and Offices to contribute occasional guest posts to the blog to highlight some of the interesting science that they support and conduct. This is the 27th in the series of NIH guest posts that will run until a new permanent NIH director is in place.
Encouraging News for Kids with Neurofibromatosis Type 1
Posted on by Dr. Francis Collins
Amid all the headlines and uncertainty surrounding the current COVID-19 pandemic, it’s easy to overlook the important progress that biomedical research is making against other diseases. So, today, I’m pleased to share word of what promises to be the first effective treatment to help young people suffering from the consequences of a painful, often debilitating genetic disorder called neurofibromatosis type 1 (NF1).
This news is particularly meaningful to me because, 30 years ago, I led a team that discovered the gene that underlies NF1. About 1 in 3,000 babies are born with NF1. In about half of those affected, a type of tumor called a plexiform neurofibroma arises along nerves in the skin, face, and other parts of the body. While plexiform neurofibromas are not cancerous, they grow steadily and can lead to severe pain and a range of other health problems, including vision and hearing loss, hypertension, and mobility issues.
The good news is the results of a phase II clinical trial involving NF1, just published in the New England Journal of Medicine. The trial was led by Brigitte Widemann and Andrea Gross, researchers in the Center for Cancer Research at NIH’s National Cancer Institute.
The trial’s results confirm that a drug originally developed to treat cancer, called selumetinib, can shrink inoperable tumors in many children with NF1. They also establish that the drug can help affected kids make significant improvements in strength, range of motion, and quality of life. While selumetinib is not a cure, and further studies are still needed to see how well the treatment works in the long term, these results suggest that the first effective treatment for NF1 is at last within our reach.
Selumetinib blocks a protein in human cells called MEK. This protein is involved in a major cellular pathway known as RAS that can become dysregulated and give rise to various cancers. By blocking the MEK protein in animal studies and putting the brakes on the RAS pathway when it malfunctions, selumetinib showed great initial promise as a cancer drug.
Selumetinib was first tested several years ago in people with a variety of other cancers, including ovarian and non-small cell lung cancers. The clinical research looked good at first but eventually stalled, and so did much of the initial enthusiasm for selumetinib.
But the enthusiasm picked up when researchers considered repurposing the drug to treat NF1. The neurofibromas associated with the condition were known to arise from a RAS-activating loss of the NF1 gene. It made sense that blocking the MEK protein might blunt the overactive RAS signal and help to shrink these often-inoperable tumors.
An earlier phase 1 safety trial looked promising, showing for the first time that the drug could, in some cases, shrink large NF1 tumors [2]. This fueled further research, and the latest study now adds significantly to that evidence.
In the study, Widemann and colleagues enrolled 50 children with NF1, ranging in age from 3 to 17. Their tumor-related symptoms greatly affected their wellbeing and ability to thrive, including disfigurement, limited strength and motion, and pain. Children received selumetinib alone orally twice a day and went in for assessments at least every four months.
As of March 2019, 35 of the 50 children in the ongoing study had a confirmed partial response, meaning that their tumors had shrunk by more than 20 percent. Most had maintained that response for a year or more. More importantly, the kids also felt less pain and were more able to enjoy life.
It’s important to note that the treatment didn’t work for everyone. Five children stopped taking the drug due to side effects. Six others progressed while on the drug, though five of them had to reduce their dose because of side effects before progressing. Nevertheless, for kids with NF1 and their families, this is a big step forward.
Drug developer AstraZeneca, working together with the researchers, has submitted a New Drug Application to the Food and Drug Administration (FDA). While they’re eagerly awaiting the FDA’s decision, the work continues.
The researchers want to learn much more about how the drug affects the health and wellbeing of kids who take it over the long term. They’re also curious whether it could help to prevent the growth of large tumors in kids who begin taking it earlier in the course of the disease, and whether it might benefit other features of the disorder. They will continue to look ahead to other potentially promising treatments or treatment combinations that may further help, and perhaps one day even cure, kids with NF1. So, even while we cope with the COVID-19 pandemic, there are reasons to feel encouraged and grateful for continued progress made throughout biomedical research.
References:
[1] Selumitinib in children with inoperable plexiform neurofibromas. New England Journal of Medicine. Gross AM, Wolters PL, Dombi E, Baldwin A, Whitcomb P, Fisher MJ, Weiss B, Kim A, Bornhorst M, Shah AC, Martin S, Roderick MC, Pichard DC, Carbonell A, Paul SM, Therrien J, Kapustina O, Heisey K, Clapp DW, Zhang C, Peer CJ, Figg WD, Smith M, Glod J, Blakeley JO, Steinberg SM, Venzon DJ, Doyle LA, Widemann BC. 18 March 2020. N Engl J Med. 2020 Mar 18. [Epub ahead of publication.]
[2] Activity of selumetinib in neurofibromatosis type 1-related plexiform neurofibromas. Dombi E, Baldwin A, Marcus LJ, Fisher MJ, Weiss B, Kim A, Whitcomb P, Martin S, Aschbacher-Smith LE, Rizvi TA, Wu J, Ershler R, Wolters P1, Therrien J, Glod J, Belasco JB, Schorry E, Brofferio A, Starosta AJ, Gillespie A, Doyle AL, Ratner N, Widemann BC. N Engl J Med. 2016 Dec 29;375(26):2550-2560.
Links:
Neurofibromatosis Fact Sheet (National Institute of Neurological Disorders and Stroke/NIH)
Brigitte Widemann (National Cancer Institute/NIH)
Andrea Gross (National Cancer Institute/NIH)
NIH Support: National Cancer Institute
Working to Improve Immunotherapy for Lung Cancer
Posted on by Dr. Francis Collins

For those who track cancer statistics, this year started off on a positive note with word that lung cancer deaths continue to decline in the United States [1]. While there’s plenty of credit to go around for that encouraging news—and continued reduction in smoking is a big factor—some of this progress likely can be ascribed to a type of immunotherapy, called PD-1 inhibitors. This revolutionary approach has dramatically changed the treatment landscape for the most common type of lung cancer, non-small cell lung cancer (NSCLC).
PD-1 inhibitors, which have only been available for about five years, prime one component of a patient’s own immune system, called T cells, to seek and destroy malignant cells in the lungs. Unfortunately, however, only about 20 percent of people with NSCLC respond to PD-1 inhibitors. So, many researchers, including the team of A. McGarry Houghton, Fred Hutchinson Cancer Research Center, Seattle, are working hard to extend the benefits of immunotherapy to more cancer patients.
The team’s latest paper, published in JCI Insight [2], reveals that one culprit behind a poor response to immunotherapy may be the immune system’s own first responders: neutrophils. Billions of neutrophils circulate throughout the body to track down abnormalities, such as harmful bacteria and malignant cells. They also contact other parts of the immune system, including T cells, if help is needed to eliminate the health threat.
In their study, the Houghton team, led by Julia Kargl, combined several lab techniques to take a rigorous, unbiased look at the immune cell profiles of tumor samples from dozens of NSCLC patients who received PD-1 inhibitors as a frontline treatment. The micrographs above show tumor samples from two of these patients.
In the image on the left, large swaths of T cells (light blue) have infiltrated the cancer cells (white specks). Interestingly, other immune cells, including neutrophils (magenta), are sparse.
In contrast, in the image on the right, T cells (light blue) are sparse. Instead, the tumor teems with other types of immune cells, including macrophages (red), two types of monocytes (yellow, green), and, most significantly, lots of neutrophils (magenta). These cells arise from myeloid progenitor cells in the bone marrow, while T cells arise from the marrow’s lymphoid progenitor cell.
Though the immune profiles of some tumor samples were tough to classify, the researchers found that most fit neatly into two subgroups: tumors showing active levels of T cell infiltration (like the image on the left) or those with large numbers of myeloid immune cells, especially neutrophils (like the image on the right). This dichotomy then served as a reliable predictor of treatment outcome. In the tumor samples with majority T cells, the PD-1 inhibitor worked to varying degrees. But in the tumor samples with predominantly neutrophil infiltration, the treatment failed.
Houghton’s team has previously found that many cancers, including NSCLC, actively recruit neutrophils, turning them into zombie-like helpers that falsely signal other immune cells, like T cells, to stay away. Based on this information, Houghton and colleagues used a mouse model of lung cancer to explore a possible way to increase the success rate of PD-1 immunotherapy.
In their mouse experiments, the researchers found that when PD-1 was combined with an existing drug that inhibits neutrophils, lung tumors infiltrated with neutrophils were converted into tumors infiltrated by T cells. The tumors treated with the combination treatment also expressed genes associated with an active immunotherapy response.
This year, January brought encouraging news about decreasing deaths from lung cancer. But with ongoing basic research, like this study, to tease out the mechanisms underlying the success and failure of immunotherapy, future months may bring even better news.
References:
[1] Cancer statistics, 2020. Siegel RL, Miller KD, Jemal A. CA Cancer J Clin. 2020 Jan;70(1):7-30.
[2] Neutrophil content predicts lymphocyte depletion and anti-PD1 treatment failure in NSCLC. Kargl J, Zhu X, Zhang H, Yang GHY, Friesen TJ, Shipley M, Maeda DY, Zebala JA, McKay-Fleisch J, Meredith G, Mashadi-Hossein A, Baik C, Pierce RH, Redman MW, Thompson JC, Albelda SM, Bolouri H, Houghton AM. JCI Insight. 2019 Dec 19;4(24).
[3] Neutrophils dominate the immune cell composition in non-small cell lung cancer. Kargl J, Busch SE, Yang GH, Kim KH, Hanke ML, Metz HE, Hubbard JJ, Lee SM, Madtes DK, McIntosh MW, Houghton AM. Nat Commun. 2017 Feb 1;8:14381.
Links:
Non-Small Cell Lung Cancer Treatment (PDQ®)–Patient Version (National Cancer Institute/NIH)
Spotlight on McGarry Houghton (Fred Hutchinson Cancer Research Center, Seattle)
Houghton Lab (Fred Hutchinson Cancer Research Center)
NIH Support: National Cancer Institute
KRAS Targeted Cancer Strategy Shows Early Promise
Posted on by Dr. Francis Collins
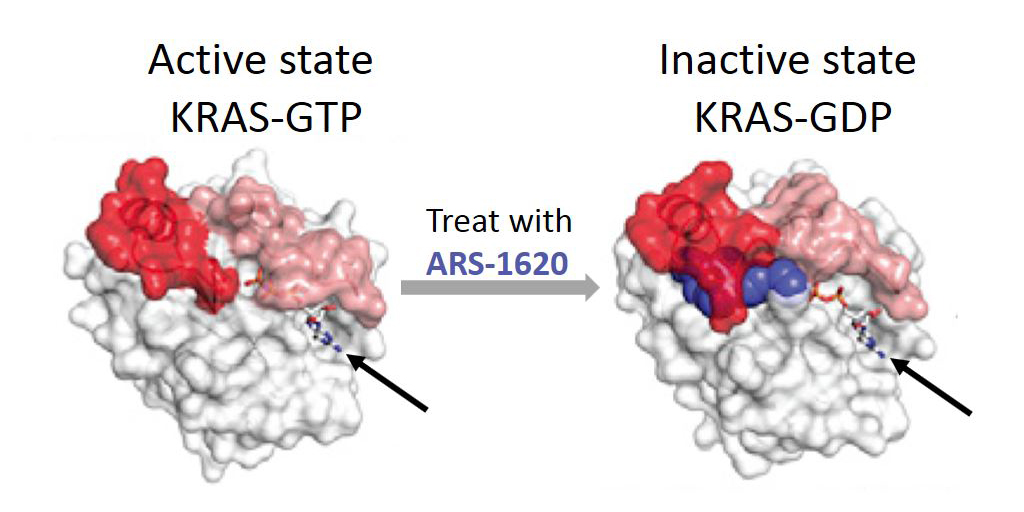
Caption: Mutant KRAS protein (white) keeps switch (red/pink) open in active state for GTP (arrow). After treatment with ARS-1620 (blue), switch is trapped in inactive GDP-bound state.
Credit: Adapted from Cell. 2018 Jan 25;172(3):578-589.
Of the more than 1.7 million Americans expected to be diagnosed with cancer this year, nearly one-third will have tumors that contain at least one mutation in the RAS family of genes [1]. That includes 95 percent of pancreatic cancers and 45 percent of colon cancers. These mutations result in the production of defective proteins that can drive cancer’s uncontrolled growth, as well as make cancers resistant to therapies. As you might expect, RAS has emerged as a major potential target for fighting cancer. Unfortunately, it is a target that’s proven very difficult to “hit” despite nearly three decades of work by researchers in both the private and public sectors, leading NIH’s National Cancer Institute to begin The RAS Initiative in 2013. This important effort has made advances with RAS that have translational potential.
Recently, I was excited to hear of progress in targeting a specific mutant form of KRAS, which is a protein encoded by a RAS gene involved in many lung cancers and some pancreatic and colorectal cancers. The new study, carried out by a pharmaceutical research team in mouse models of human cancer, is the first to show that it is possible to shrink a tumor in a living creature by directly inhibiting mutant KRAS protein [2].