self-healing
Regenerative Medicine: New Clue from Fish about Healing Spinal Cord Injuries
Posted on by Dr. Francis Collins
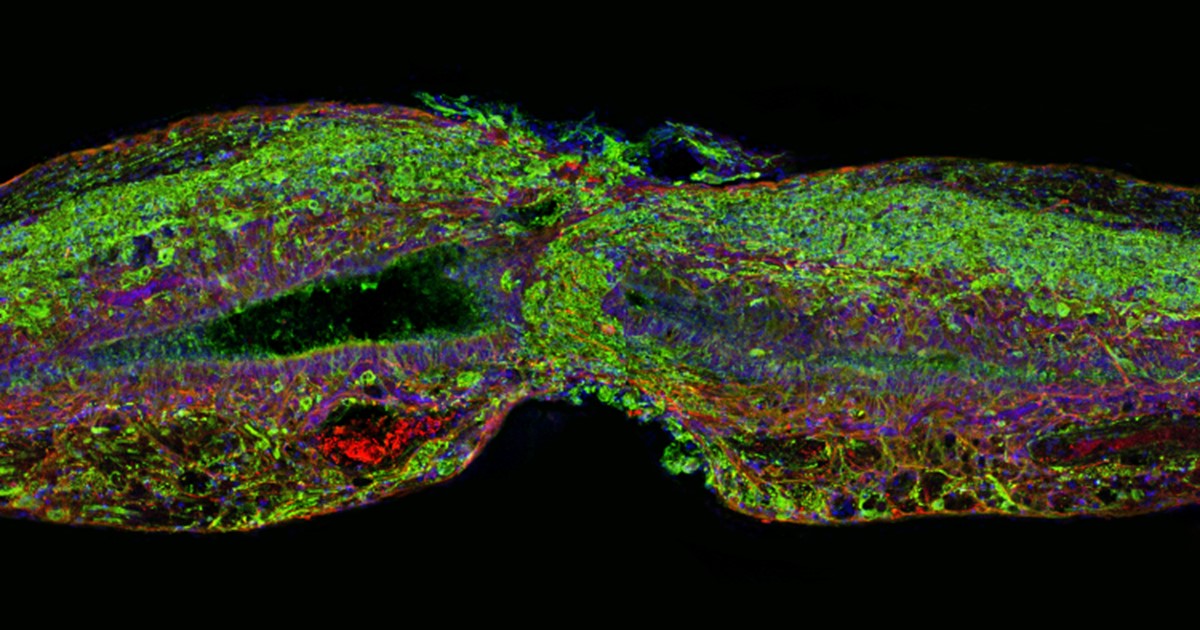
Caption: Tissue section of zebrafish spinal cord regenerating after injury. Glial cells (red) cross the gap between the severed ends first. Neuronal cells (green) soon follow. Cell nuclei are stained blue and purple.
Credit: Mayssa Mokalled and Kenneth Poss, Duke University, Durham, NC
Certain organisms have remarkable abilities to achieve self-healing, and a fascinating example is the zebrafish (Danio rerio), a species of tropical freshwater fish that’s an increasingly popular model organism for biological research. When the fish’s spinal cord is severed, something remarkable happens that doesn’t occur in humans: supportive cells in the nervous system bridge the gap, allowing new nerve tissue to restore the spinal cord to full function within weeks.
Pretty incredible, but how does this occur? NIH-funded researchers have just found an important clue. They’ve discovered that the zebrafish’s damaged cells secrete a molecule known as connective tissue growth factor a (CTGFa) that is essential in regenerating its severed spinal cord. What’s particularly encouraging to those looking for ways to help the 12,000 Americans who suffer spinal cord injuries each year is that humans also produce a form of CTGF. In fact, the researchers found that applying human CTGF near the injured site even accelerated the regenerative process in zebrafish. While this growth factor by itself is unlikely to produce significant spinal cord regeneration in human patients, the findings do offer a promising lead for researchers pursuing the next generation of regenerative therapies.
Lab-Grown Muscle Bundles: A Glimpse of the Future?
Posted on by Dr. Francis Collins

Caption: Engineered muscle fibers are stained with red and green dyes that recognize particular protein markers. The yellow color results from a combination of red and green. The blue dots are cell nuclei.
Credit: Duke University
When you do a hard workout at the gym, or run a marathon, you generate lots of little tears in muscle. This is usually not a problem and may even lead to improved muscle strength—because the injury activates stem cells in the muscle (called satellite cells) that replicate and form new muscle fibers to repair and rebuild the damaged tissue. But when injuries extend beyond the normal wear and tear—a major injury or resection, for example—this amazing self-healing system isn’t enough. That’s when a self-healing, lab-grown muscle transplant would be particularly useful—but we haven’t yet been able to create this in a dish.
