Rare Disease Mystery: Nodding Syndrome May Be Linked to Parasitic Worm
Posted on by Dr. Francis Collins

Caption: Village in the East Africa nation of Uganda
Credit: Centers for Disease Control and Prevention
In the early 1960s, reports began to surface that some children living in remote villages in East Africa were suffering mysterious episodes of “head nodding.” The condition, now named nodding syndrome, is recognized as a rare and devastating form of epilepsy. There were hints that the syndrome might be caused by a parasitic worm called Onchocerca volvulus, which is transmitted through the bites of blackflies. But no one had been able to tie the parasitic infection directly to the nodding heads.
Now, NIH researchers and their international colleagues think they’ve found the missing link. The human immune system turns out to be a central player. After analyzing blood and cerebrospinal fluid of kids with nodding syndrome, they detected a particular antibody at unusually high levels [1]. Further studies suggest the immune system ramps up production of that antibody to fight off the parasite. The trouble is those antibodies also react against a protein in healthy brain tissue, apparently leading to progressive cognitive dysfunction, neurological deterioration, head nodding, and potentially life-threatening seizures.
The findings, published in Science Translational Medicine, have important implications for the treatment and prevention of not only nodding syndrome, but perhaps other autoimmune-related forms of epilepsy. As people in the United States and around the globe today observe the 10th anniversary of international Rare Disease Day, this work provides yet another example of how rare disease research can shed light on more common diseases and fundamental aspects of human biology.
Avindra Nath of NIH’s National Institute of Neurological Disorders and Stroke first learned of nodding syndrome at a meeting in Uganda in 2012. In some parts of the country, half of families had a child affected by the condition, leaving many kids in need of constant supervision and medical care. He also heard the condition was a problem in Tanzania and South Sudan.
In search of a cause, Nath was told, scientists had already looked to the immune system, genetics, toxins, nutritional factors, and more. But they still didn’t have an answer. Intrigued, Nath agreed to help.
Nath and his colleagues knew that kids with nodding syndrome often showed signs of onchocerciasis, a devastating skin disease caused by the worm O. volvulus. Onchocerciasis can lead to “river blindness” as the worms migrate into the eyes. But it wasn’t clear how this parasitic infection could affect the brain. Nath, along with his former postdoc Tory Johnson, now at Johns Hopkins School of Medicine, Baltimore, and Thomas Nutman, an onchocerciasis researcher at NIH’s National Institute of Allergy and Infectious Diseases, suspected the problem was autoimmune, a situation in which the body’s defenses accidentally attack its own cells and tissues.
Knowing that earlier attempts to implicate the immune system had come up empty, Johnson decided to take a new approach. She used more sophisticated protein chip technology. The new approach allowed her to screen for thousands of antibodies at once in pooled samples from kids with nodding syndrome and compare them to those of healthy controls.
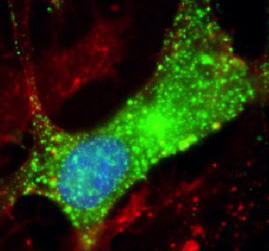
Caption: Leiomodin-1 (green) inside human neurons.
Blue is the nucleus; red indicates the cell membrane.
Credit: Avindra Nath, M.D., National Institute of Neurological
Disorders and Stroke
When Johnson saw the results, one thing immediately jumped out. Antibodies in the blood of youngsters with nodding syndrome reacted 33,000 times more strongly against a protein called leiomodin-1 than in unaffected controls. While the leiomodin-1 antibodies turned up in members of both groups, they seemed to reach much higher levels in many people with nodding syndrome. What’s more, the antibodies were present in cerebrospinal fluid from the central nervous system—so they had somehow crossed the blood-brain barrier.
At the time, Johnson and Nath had never heard of leiomodin-1. A search of the scientific literature indicated the protein was first characterized only about five years ago [2] and is found primarily in muscle and thyroid. But there was no mention of the central nervous system.
The researchers went on to show that leiomodin-1 is actually expressed in mature and developing human neurons. In mouse studies, they also found leiomodin-1 in the very areas of the brain that appear to be affected in humans with nodding syndrome.
As additional evidence, they showed that blood from kids with nodding syndrome containing those leiomodin-1 antibodies could kill neurons in lab dishes. Neurons treated with blood depleted of the leiomodin-1 antibodies survived.
Why would infection with O. volvulus lead to overproduction of neurotoxic leiomodin-1 antibodies? It turns out the worms produce proteins with structural similarities to leiomodin-1. Indeed, the researchers show, leiomodin-1 antibodies also react against several proteins found in the worms’ bodies.
The findings, however, aren’t yet an open-and-shut case. More research is needed to nail down the cause and effect of the infection. But what the evidence does show is once the antibodies attack neurons, the damage is likely permanent. The best way to prevent nodding syndrome is to keep people from becoming infected with O. volvulus in the first place. In fact, efforts to curb the parasitic infection in Uganda with the drug ivermectin (Mectizan®) have already produced a drastic reduction in the incidence of nodding syndrome.
Now that this mystery has been unraveled, it’s tempting to speculate that nodding syndrome may not be the only form of autoimmune-related epilepsy. In fact, in most cases the causes of those conditions still aren’t known. These new findings should encourage researchers to use more powerful technologies and take another hard look at the antibodies and infectious agents—bacteria, viruses, or parasites—that might be responsible for other forms of epilepsy.
References:
[1] Nodding syndrome may be an autoimmune reaction to the parasitic worm Onchocerca volvulus. Johnson TP, Tyagi R, Lee PR, Lee MH, Johnson KR, Kowalak J, Elkahloun A, Medynets M, Hategan A, Kubofcik J, Sejvar J, Ratto J, Bunga S, Makumbi I, Aceng JR, Nutman TB, Dowell SF, Nath A. Sci Transl Med. 2017 Feb 15;9(377).
[2] Leiomodin 1, a new serum response factor-dependent target gene expressed preferentially in differentiated smooth muscle cells. Nanda V, Miano JM. J Biol Chem. 2012 Jan 20;287(4):2459-2467.
Links:
Nodding Syndrome (Genetic and Rare Diseases Information Center/National Center for Advancing Translational Sciences/NIH)
Nodding Syndrome (World Health Organization)
Onchocerciasis (World Health Organization)
Avindra Nath (National Institute of Neurological Disorders and Stroke/NIH)
Tory Johnson (Johns Hopkins School of Medicine, Baltimore, MD)
Thomas Nutman (National Institute of Allergy and Infectious Diseases/NIH)
NIH Support: National Institute of Neurological Disorders and Stroke; National Institute of Allergy and Infectious Diseases; National Human Genome Research Institute
