Mouse Study Finds Microbe Might Protect against Food Poisoning
Posted on by Dr. Francis Collins
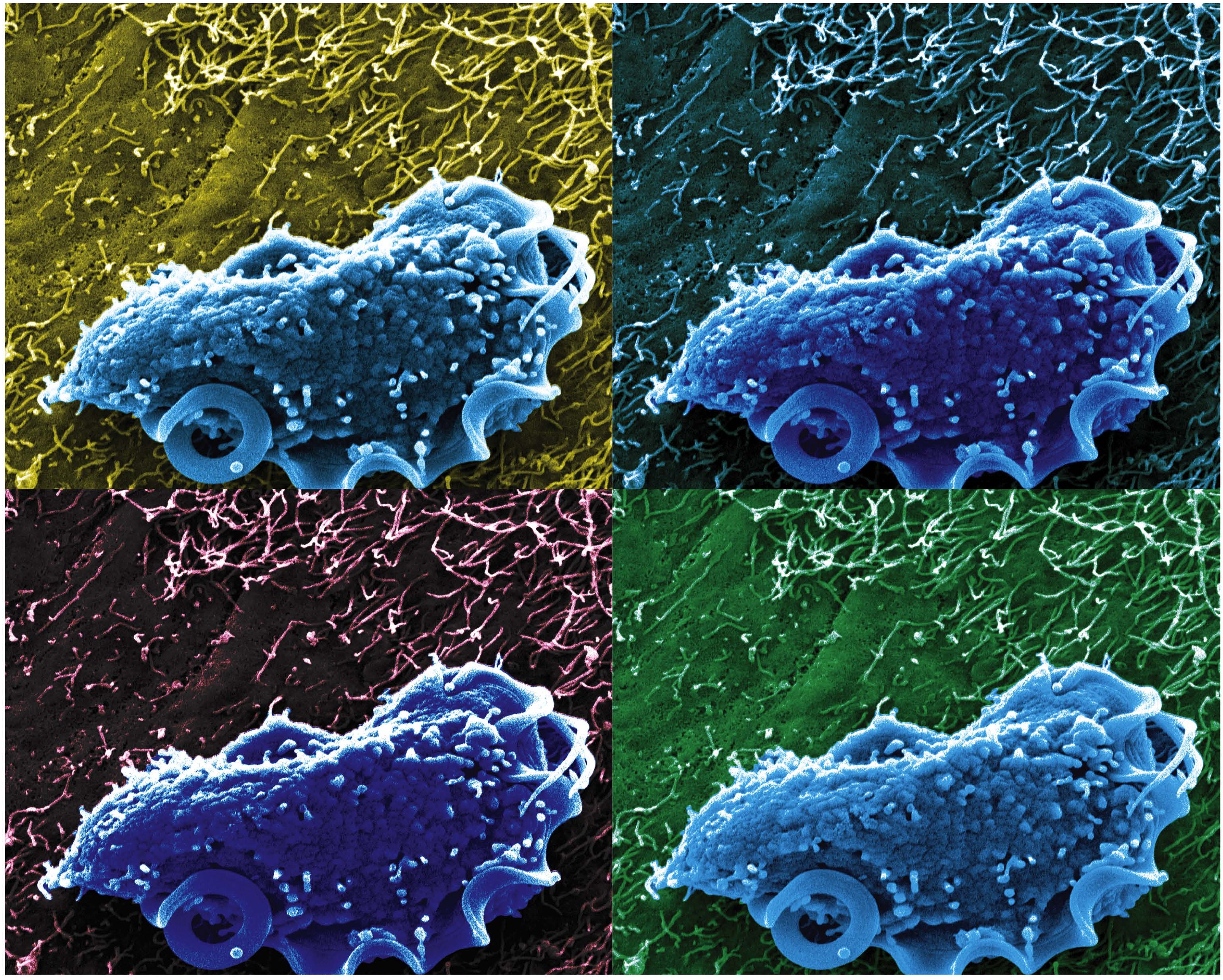
Caption: Scanning electron microscopy image of T. mu in the mouse colon.
Credit: Aleksey Chudnovskiy and Miriam Merad, Icahn School of Medicine at Mount Sinai
Recently, we humans have started to pay a lot more attention to the legions of bacteria that live on and in our bodies because of research that’s shown us the many important roles they play in everything from how we efficiently metabolize food to how well we fend off disease. And, as it turns out, bacteria may not be the only interior bugs with the power to influence our biology positively—a new study suggests that an entirely different kingdom of primarily single-celled microbes, called protists, may be in on the act.
In a study published in the journal Cell, an NIH-funded research team reports that it has identified a new protozoan, called Tritrichomonas musculis (T. mu), living inside the gut of laboratory mice. That sounds bad—but actually this little wriggler was potentially providing a positive benefit to the mice. Not only did T. mu appear to boost the animals’ immune systems, it spared them from the severe intestinal infection that typically occurs after eating food contaminated with toxic Salmonella bacteria. While it’s not yet clear if protists exist that can produce similar beneficial effects in humans, there is evidence that a close relative of T. mu frequently resides in the intestines of people around the world.
The discovery comes from Miriam Merad at the Icahn School of Medicine at Mount Sinai, New York, and colleagues, including graduate student Aleksey Chudnovskiy and postdoctoral fellow Arthur Mortha. Their study started out by accident after the team members noticed something unusual: mice bred at their facility had more of certain types of immune cells in their guts than mice purchased from a commercial vendor.
To find out why, they examined fecal material from both groups of mice under a scanning electron microscope. Their studies uncovered unfamiliar single-celled protozoa with whip-like tails in samples taken from the guts of their Mount Sinai mice and not in those from the other lab.
Michael Grigg, a molecular parasitologist at the NIH’s National Institute of Allergy and Infectious Diseases, sequenced the DNA of this mysterious organism and found it was a species of protozoa that had never been described before. Further work showed it was closely related genetically to a known protozoan parasite found in mice. Now curious, Grigg decided to check the mice from four NIH animal facilities. They carried T. mu.
The Mount Sinai researchers went on to show that T. mu readily takes up residence in the guts of mice without causing them any immediate harm. Closer observation also showed that T. mu’s introduction led to a rapid increase in immune cells, primarily dendritic and T cells, just as was seen in the Mount Sinai mice.
Further study showed that T. mu activates cellular machinery that produces inflammation to generate that heightened immune response. What’s more, it appears that this immune boost is beneficial. When the researchers infected mice with the bacterium Salmonella typhimurium, a leading cause of food-borne infection in developing countries, those with T. mu in their guts showed almost no signs of illness. For those without T. mu, the Salmonella infection led to massive intestinal inflammation and distress.
But this protozoan isn’t always beneficial. The researchers found that T. mu made mice with preexisting inflammatory disease sicker, as evidenced by increased weight loss and tissue damage. T. mu also led to an increase in the number of tumors in the guts of mice with a genetic susceptibility for colon cancer.
To explore whether the findings in mice could possibly have implications for humans, Grigg screened 188 fecal samples collected from healthy people in Colombia in search of a related organism. Those studies turned up DNA evidence of a related protozoan called Dientamoeba fragilis (D. fragilis) in the guts of many people. That finding was confirmed in another set of 96 human samples taken from people around the world, including other parts of South America, Africa, Europe, and Asia.
Up until now, protists have mainly been recognized and studied as disease-causing parasites. Familiar examples include Giardia, which people contract from drinking contaminated water from outdoor sources, and Toxoplasma gondii, which people can get from their pet cats. The T. mu relative D. fragilis has been noted for its links to irritable bowel syndrome (IBD), although its effects on patients with IBD—for better or worse—aren’t yet clear.
Based on the new findings, Merad and Chudnovskiy say it now appears that protists probably aren’t all bad when it comes to our health. As in the laboratory mice, they may afford us with extra immune protection, which could be especially beneficial for those living in parts of the world where infectious disease is an ever-present threat.
It’s possible also that an absence of such protists could explain why some people are more susceptible to food poisoning or other infections than others. Whether the evidence ultimately bears that out or not, the time has apparently come to give more attention to the protists that make up part of the human microbiome.
Reference:
[1] Host-protozoan interactions protect from muscosal infections through activation of the inflammasome. Chudnovskiy A, Mortha A, Kana V, Kennard A, Ramirez JD, Rahman A, Remark R, Mogno I, Ng R, Gnjatic S, Amir ED, Solovyov A, Greenbaum B, Clemente J, Faith J, Belkaid Y, Grigg ME, Merad M. Cell. 2016 Oct 6;167(2):444-456.
Links:
Human Microbiome Project (Common Fund/NIH)
Miriam Merad (Mount Sinai, New York City)
NIH Support: National Institute of Allergy and Infectious Diseases; National Cancer Institute

Toxoplasmosis is caused by Toxoplasma gondii, a parasite infection. It’s one of the most prevalent parasites that infect humans and animals when they eat spoiled or badly cooked meat. Furthermore, the disease can be passed from mother to infant during pregnancy and through contact with contaminated cat feces. Toxoplasma infection affects more than 60% of the world’s population and is a major public health concern.