Saving Fat for Lean Times
Posted on by Lawrence Tabak, D.D.S., Ph.D.
Humans and all multi-celled organisms, or metazoans, have evolved through millennia into a variety of competing shapes, sizes, and survival strategies. But all metazoans still share lots of intriguing cell biology, including the ability to store excess calories as fat. In fact, many researchers now consider fat-storing cells to be “nutrient sinks,” or good places for the body to stash excess sugars and lipids. Not only can these provide energy needed to survive a future famine, this is a good way to sequester extra molecules that could prove toxic to cells and organs.
Here’s something to think about the next time you skip a meal. Fat-storing cells organize their fat reserves spatially, grouping them into specific pools of lipid types, in order to generate needed energy when food is scarce.
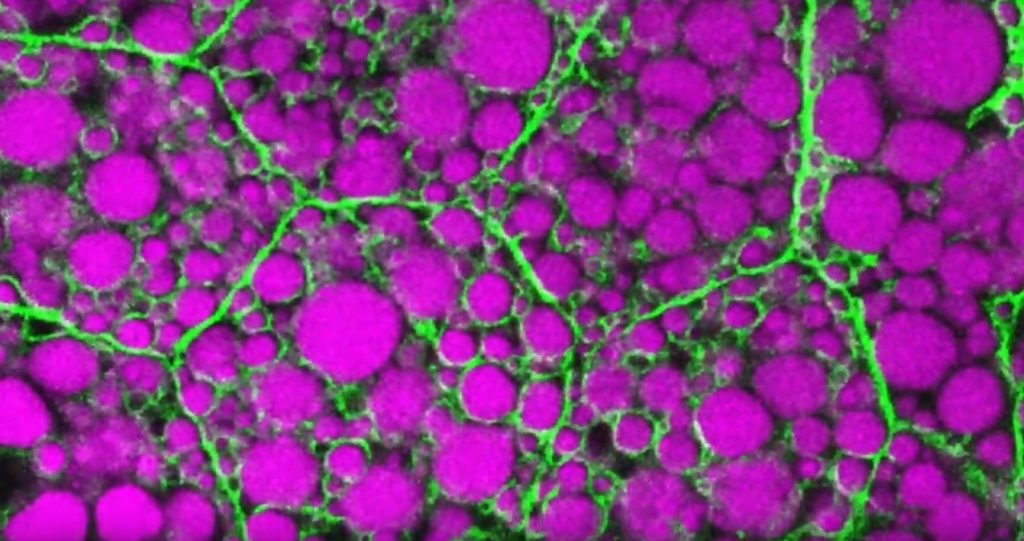
That’s the story behind this striking image taken in a larval fruit fly (Drosophila melanogaster). The image captures fat-storing adipocytes in an organ called a fat body, where a larval fruit fly stores extra nutrients. It’s like the fat tissue in mammals. You can see both large and small lipid droplets (magenta) inside polygon-shaped fat cells, or adipocytes, lined by their plasma membranes (green). But notice that the small lipid droplets are more visibly lined by green, as only these are destined to be saved for later and exported when needed into the fly’s bloodstream.
Working in Mike Henne’s lab at the University of Texas Southwestern Medical Center, Dallas, research associate Rupali Ugrankar discovered how this clever fat-management system works in Drosophila [1]. After either feeding flies high-or-extremely low-calorie diets, Ugrankar used a combination of high-resolution fluorescence confocal microscopy and thin-section transmission electron microscopy to provide a three-dimensional view of adipocytes and their lipid droplets inside.
She observed two distinct sizes of lipid droplets and saw that only the small ones clustered at the cell surface membrane. The adipocytes contorted their membrane inward to grab these small droplets and package them into readily exportable energy bundles.
Ugrankar saw that during times of plenty, a protein machine could fill these small membrane-wrapped fat droplets with lots of triacylglycerol, a high-energy, durable form of fat storage. Their ready access at the surface of the adipocyte allows the fly to balance lipid storage locally with energy release into its blood in times of famine.
Ugrankar’s adeptness at the microscope resulted in this beautiful photo, which was earlier featured in the American Society for Cell Biology’s Green Fluorescent Protein Image and Video Contest. But her work and that of many others help to open a vital window into nutrition science and many critical mechanistic questions about the causes of obesity, insulin resistance, hyperglycemia, and even reduced lifespan.
Such basic research will provide the basis for better therapies to correct these nutrition-related health problems. But the value of basic science must not be forgotten—some of the most important leads could come from a tiny insect in its larval state that shares many aspects of mammalian metabolism.
Reference:
[1] Drosophila Snazarus regulates a lipid droplet population at plasma membrane-droplet contacts in adipocytes. Ugrankar R, Bowerman J, Hariri H, Chandra M, et al. Dev Cell. 2019 Sep 9;50(5):557-572.e5.
Links:
The Interactive Fly (Society for Developmental Biology, Rockville, MD)
Henne Lab (University of Texas Southwestern Medical Center, Dallas)
NIH Support: National Institute of General Medical Sciences
Share this:
- Click to share on LinkedIn (Opens in new window)
- Click to share on Pinterest (Opens in new window)
- Click to share on Tumblr (Opens in new window)
- Click to share on Reddit (Opens in new window)
- Click to share on Telegram (Opens in new window)
- Click to share on WhatsApp (Opens in new window)
- Click to print (Opens in new window)
Posted In: Snapshots of Life
Tags: 2019 Green Fluorescent Protein Image and Video Contest, adipocyte, American Society for Cell Biology, basic research, cell biology, Drosophila melanogaster, fat, fat body, fat cells, fat storage, fluorescence microscopy, fruit fly, hyperglycemia, imaging, insulin resistance, lifespan, lipid droplet, lipid storage, lipids, metabolism, model organisms, nutrient sink, obesity, triacylglycerol

Beautiful picture. It makes a very useful talking point when explaining how adipocytes are dynamic contributors to energy storage and utilization.
Good job Rupali👍